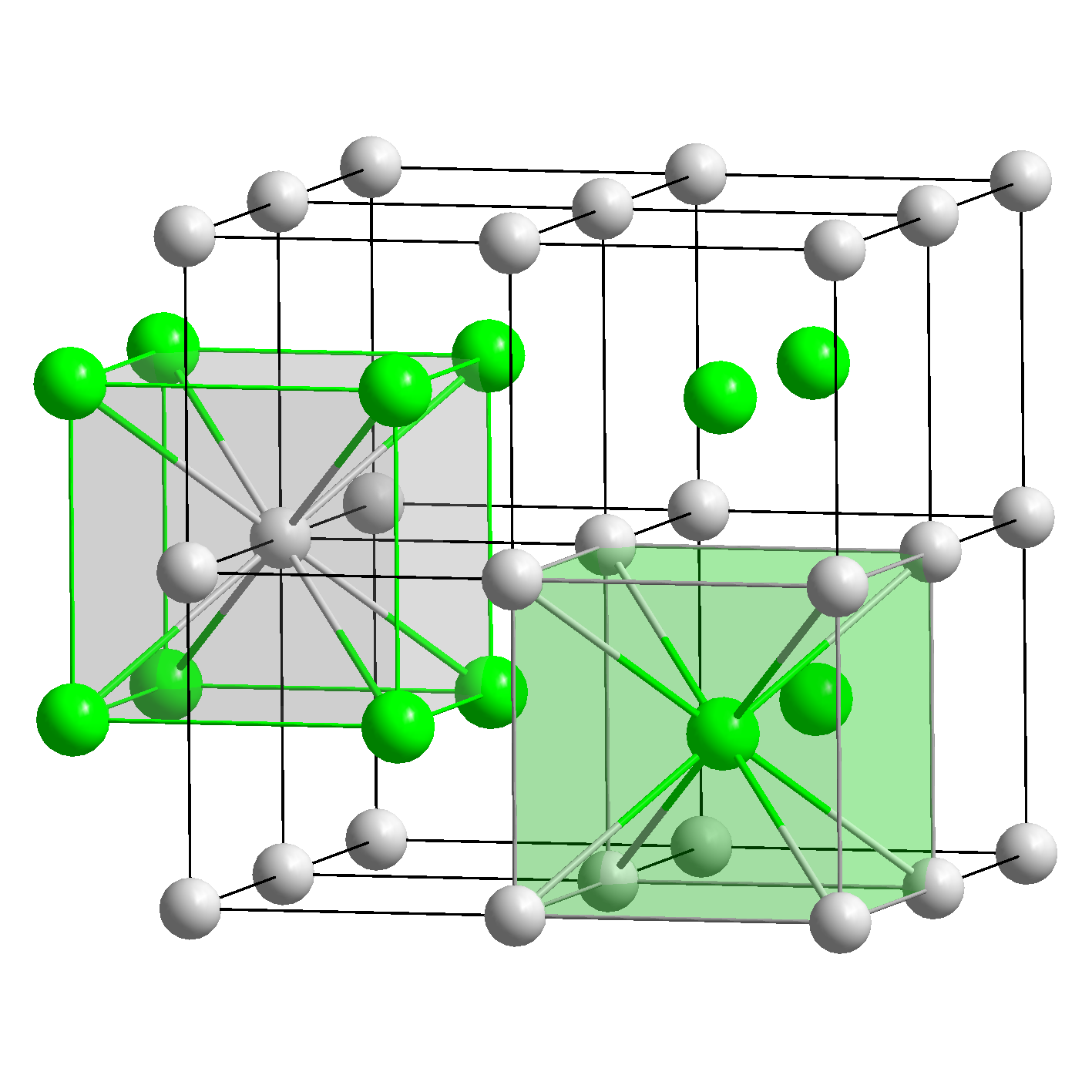
The following elements are common metals:Īluminum(Al), barium(Ba), beryllium(Be), bismuth(Bi), cadmium(Cd), calcium(Ca), cerium(Ce), cesium(Cs), chromium(Cr), cobalt(Co), copper(Cu), gold(Au), indium(In), iridium(Ir), iron(Fe), lead(Pb), lithium(Li), magnesium(Mg), manganese(Mn), mercury(Hg), molybdenum(Mo), nickel(Ni), osmium(Os), palladium(Pd), platinum(Pt), potassium(K), radium(Ra), rhodium(Rh), silver(Ag), sodium(Na), tantalum(Ta), thallium(Tl), thorium(Th), tin(Sn), titanium(Ti), tungsten(W), uranium(U), vanadium(V), zinc(Zn). These properties are also associated with the metallic bonding and presence of free electrons in the crystal lattice. Devices such as solid state transistors, lasers, solar cells, and light emitting diodes are often made from single crystals. A small part of the crystal that can be repeated to form the entire crystal is called a unit cell. Other specific metal features are: luster or shine of their surface (when polished), their malleability (ability to be hammered) and ductility (ability to be drawn). The structure of a crystalline solid, whether a metal or not, is best described by considering its simplest repeating unit, which is referred to as its unit. In a crystal, atoms are arranged in straight rows in a three-dimensional periodic pattern. These free electrons belong to the whole metal crystal.Ībility of the valence free electrons to travel throughout the solid explains both the high electrical conductivity and thermal conductivity of metals. Each of the atoms of the metal contributes its valence electrons to the crystal lattice, forming an electron cloud or electron “gas”, surrounding positive metal ions. Metal crystal structure and specific metal properties are determined by metallic bonding – force, holding together the atoms of a metal. The smallest possible part of crystal lattice, determining the structure, is called primitive unit cell.Įxamples of typical crystal lattice are presented in the picture: The crystalline structure is described in terms of crystal lattice, which is a lattice with atoms or ions attached to the lattice points. A lattice point here can represent an atom or a point, depending on the context.įinally, as mentioned by other posts, these points can be vacant.Crystalline solid substance is characterized by atoms arranged in a regular pattern, extending in all three dimensions. Some of the properties of crystalline solids depend on the crystal structure of the material, the manner in which atoms, ions, or molecules are spatially arranged. There are 14 general types of such patterns known as Bravais lattices. There are seven different lattice systems, some of which have more than one type of lattice, for a total of fourteen different unit cells, which have the shapes shown in Figure 10.6.11. However, I personally think that this video explains it better. A crystal lattice is a repeating pattern of mathematical points that extends throughout space. Figure 10.6.10: A unit cell is defined by the lengths of its three axes (a, b, and c) and the angles (,, and ) between the axes. These are trigonal, hexagonal, tetrahedral, orthorhombic, monoclinic, and triclinic. More often than not, it is used to refer to the Fourier transform of the Bravais lattice. A number of heat-treatment cycles have been developed to alter the structure of steel. Lattices are classified according to their dominant symmetries. The reciprocal lattice is the Fourier transform of either the crystal lattice or a Bravais lattice. What matters in a Bravais lattice are not the points themselves, but how they are arranged (i.e. 
In other words, a lattice point in a Bravais lattice is a point, which is equal and indistinguishable from any other another point. Instead a lattice point represents a position in which an atom can be placed. When multiple graphene sheets are layered on top of each other, van der Walls bonding occurs and the three di-mensional structure of graphite is formed with a lattice FIG. Unlike the crystal lattice, however, lattice points in the Bravais lattice no longer represent a position of a particular atom. Lattice Structure Our discussion of the crystal structure of graphite fol-lows partially from D.D.L. They are pretty much the same as crystal lattices. For example, in NaCl, a lattice point in a crystal lattice represents the position of a sodium ion or a chloride ion.īravais lattices are more mathematical and abstract than crystal lattices. The crystal lattice is used to describe the lattice of a real crystal. I will also be assuming that we are dealing with crystals. Once the positions of an atom and its neighbours are known at one point, the place of each atom is known. However, I will be assuming that we are talking about periodic lattices. A solid is crystalline if it has long-range order. I am unsure whether lattices can only describe periodic systems (see quasicrystals). Lattices are used to describe highly ordered systems such as crystals and some supersolids. A lattice point is a point in any of these lattices.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
